 “The UK chemical industry is warning of a £1bn cost to duplicate EU regime. Smaller companies may fail to survive the move to a British safety registrations agency”.
“The UK chemical industry is warning of a £1bn cost to duplicate EU regime. Smaller companies may fail to survive the move to a British safety registrations agency”.
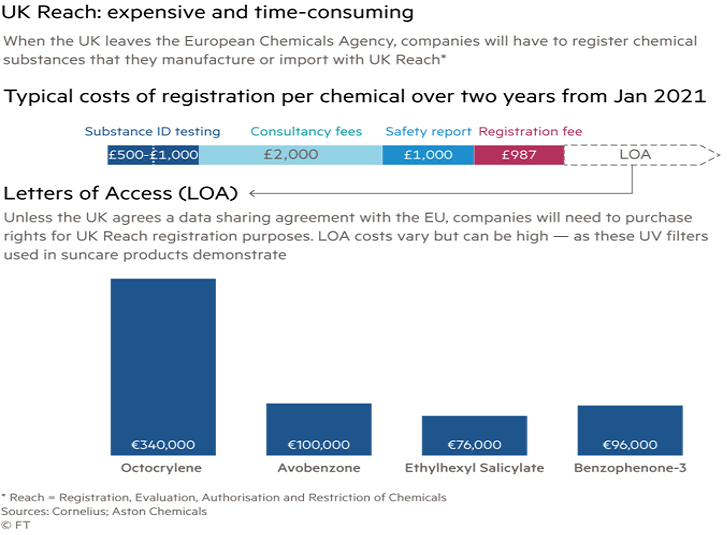
The Financial Times reports that “Registering a single chemical in the new UK Reach database could cost up to £300,000 if companies are required to buy “letters of access” to use the vast banks of test data held by ECHA — information that is expensive to produce and often owned by third parties. Even then, additional testing may be required.
“Steve Elliott, chief executive of the Chemical Industries Association, said that unless a data-sharing deal was done with Brussels the new system would add more than £1bn in costs to companies, just to duplicate existing registrations. To make matters more complex, companies has still not seen the computer software that the Health and Safety Executive will use to collect the new UK registrations. “The real question is how do you get hold of the registration data, which is held in commercial agreements,” he said. “It’s unclear and we’re getting closer and closer to the time when Brexit is going to happen.”


 “The UK chemical industry is warning of a £1bn cost to duplicate EU regime. Smaller companies may fail to survive the move to a British safety registrations agency”.
“The UK chemical industry is warning of a £1bn cost to duplicate EU regime. Smaller companies may fail to survive the move to a British safety registrations agency”.





